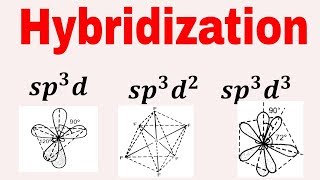
What is sp3d hybridization
The resulting orbitals are known as sp 3 hybrid orbitals. Here, the atoms do not have un-hybridized p orbitals because all three p orbitals are involved in hybridization. Sp 3 Hybridization is the mixing of one s atomic orbital with three p atomic orbitals. The atoms that undergo this hybridization have 1 un-hybridized p orbital because only two of three p orbitals are involved in this hybridization. Therefore, the angle between these orbitals is 120 oC. The spatial arrangement of sp 2 hybrid orbitals is trigonal planar. S (or p) characteristic percentage = total atomic orbitals x (1/2) x 100% Therefore the fraction of s orbital is 1/2, and the fraction of p orbitals is 1/2. The ratio between s and p orbitals is 1:1. Here, we consider that all s and p orbitals as just atomic orbitals (s+p). Therefore, after the hybridization of an s orbital with one of these p orbitals, there are two un-hybridized p orbitals present in that atom. An electron shell contains three p orbitals. Sp hybridization is the hybridization that takes place between an s atomic orbital and a p atomic orbital. Key Terms: Atomic Orbital, Hybrid Orbitals, Hybridization, Orbitals, Sp Hybridization, Sp 2 Hybridization, Sp 3 Hybridization What is the Difference Between sp sp 2 and sp 3 Hybridization – Definition, Calculation of S and P Characteristics, Other FeaturesĤ. The main difference between sp sp 2 and sp 3 hybridization is that sp hybridization forms hybrid orbitals having 50% s orbital characteristics and sp 2 hybridization forms hybrid orbitals having 33% s orbital characteristics whereas sp 3 hybridization forms hybrid orbitals having 25% s orbital characteristics. These orbitals are formed by the hybridization of s and p atomic orbitals in different ratios. There are different forms of hybridizations which make different forms of hybrid orbitals such as sp, sp 2 and sp 3 hybrid orbitals. In chemistry, hybridization is the mixing of different atomic orbitals to form hybrid orbitals. Atomic orbitals of an atom undergo hybridization to make suitable orbitals for chemical bonding. There are three main types of orbitals: atomic orbitals, molecular orbitals, and hybrid orbitals. According to different discoveries, scientists have proposed different shapes for these orbitals.

Orbitals are hypothetical structures that can be filled with electrons. Main Difference – sp vs sp 2 vs sp 3 Hybridization
